 It is a soft, silvery-gold alkali metal with a melting point of 28 ☌ (82 ☏), which makes it one of only five elemental metals that are liquid at or near room temperature. The high cost of cesium and the growing popularity of similar and cheaper technologies using other alkali metals reduced the use of cesium to a handful of applications.Caesium or cesium is a chemical element with symbol Cs and atomic number 55. In later decades, more uses for cesium arose, including photoelectric cells, spectrometers and catalysts for organic reactions.  
Cesium was used in vacuum tubes to remove traces of remaining oxygen due to its ready nature to bond with it, and as a coating on heated cathodes to increase the electric current. The first practical applications of cesium were realized in the 1920s, according to the USGS. It was discovered in 1860 by German chemists Robert Bunsen and Gustav Kirchhoff when they were analyzing the spectrum of mineral water, according to WebElements. Most common isotopes: Cs-133 (100 percent of natural abundance) HistoryĬesium was the first element to be discovered with a spectroscope. There are also at least 39 artificial isotopes created in a lab. Temperatures Melting point: 301.59 28.44 C (83.19 F) K Boiling point: 944 671 C (1240 F) K Liquid range: 642.41 K Critical temperature: 1938 1665. Number of natural isotopes (atoms of the same element with a different number of neutrons): 1. Melting point: 83.1 degrees Fahrenheit (28.4 degrees Celsius) Just the factsĪtomic number (number of protons in the nucleus): 55Ītomic symbol (on the periodic table of elements): CsĪtomic weight (average mass of the atom): 132.90ĭensity: 1.086 ounces per cubic inch (1.879 grams per cubic centimeter) Cesium is a monoisotopic element, and hence the only naturally occurring nuclide 133Cs. Cesium salts color the nonluminous flame of a Bunsen gas burner reddish violet or bluish purple (460 nm). Cesium is the most reactive of the alkali metals with nitrogen, carbon, and hydrogen. Like the other alkali metals it combines vigorously with mercury, forming a stable cesium amalgam. Moreover, cesium hydroxide is the strongest base known. It is readily soluble in liquid and anhydrous ammonia, giving deep blue solutions. Therefore, it must be kept immersed in mineral oil. 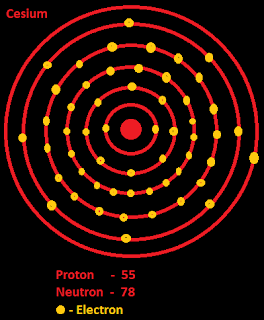
It ignites spontaneously in oxygen and hence is difficult to handle because it reacts spontaneously in air. It decomposes water vigorously, evolving hydrogen and forming the caustic cesium hydroxide, CsOH, and it even reacts with ice at temperatures above –116☌. 28.4☌), it can be liquid at room temperature. Like the other alkali metals, it has a body-centered cubic crystal lattice structure. Caesium is a soft, silvery-gold alkali metal with a melting point of 28.5 ☌, which makes it one of only five elemental metals that are liquid at or near room temperature. It would melt in your hands - if it didn't explode first, as it is highly reactive to moisture.Ĭesium is a soft, malleable, silvery-white element when freshly cut and has the highest density (1873 kg.m–3) of the alkali metals, just slightly greater than that of magnesium. Cesium is a rare, silver-white, shiny metal with brilliant blue spectral lines the element's name comes from "caesius," a Latin word meaning "sky blue." It is the softest metal, with a consistency of wax at room temperature. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
